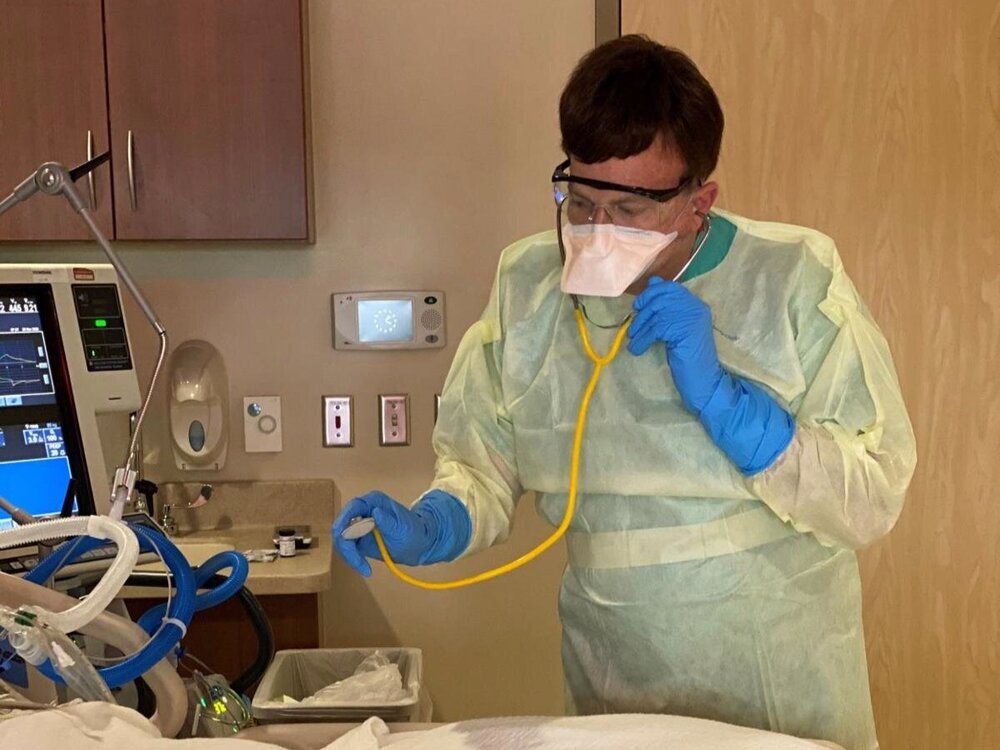
Last week, Williamson Medical Center and Vanderbilt University Medical Center became the first medical centers in the state selected to participate in a new COVID-19 prevention drug trial, dubbed the HERO trial.
That program officially launched at both medical centers Monday, and with its launch came the urging of Williamson Medical Center staff for community members to participate.
What is the HERO trial?
An acronym for Healthcare Worker Exposure Response and Outcomes, the HERO trial is a federal research program aimed at determining the effectiveness of plaquenil hydroxychloroquine in preventing the contraction of COVID-19. The drug had previously been touted by President Donald Trump as showing “great promise” in fighting the growing number of COVID-19 cases.
The HERO trial will see a total of 15,000 health care workers and first responders enrolled across 40 different sites across the country, with Williamson Medical Center and Vanderbilt being two of them. Over a month-long period, participants will be given plaquenil hydroxychloroquine, after which researches will determine its effectiveness in preventing the contraction of COVID-19.
The trial is in part conducted by the National Patient-Centered Clinical Research Network (PCORnet), a national patient data network that aims to improve the capacity to conduct comparative effectiveness research.
A call to action for the Williamson County community
Aaron Milstone, the principal investigator for the HERO trial at Williamson Medical, is now calling on Williamson County residents to consider participating in the trial, whether it be as an enrollee or by taking surveys.
Those eligible to participate in the trial include nurses, therapists, physicians, emergency responders, food service workers, environmental services workers, interpreters, transporters, or anyone working in a setting where people receive health care.
“I’m calling on my fellow Williamson County healthcare workers to share their perspectives so that we can understand and provide answers to the problems they face in real time – and over time,” Milstone said. “We have long been working with PCORnet to answer big healthcare questions together. COVID-19 is the challenge we are facing today and PCORnet is ready to meet the moment.”
Those participating in the HERO trial who are open to taking plaquenil hydroxychloroquine will either be given the drug or a placebo over a month-long period. Rates of COVID-19 infection would then be compared between the two groups at the conclusion of the trial to measure the drug’s effectiveness.
Health care workers not willing to take plaquenil hydroxychloroquine can still help by taking surveys.
“It is hard to think that by doing my daily job, not only can I get sick, but I can infect those that I love the most,” said Jorge Lascano, associate professor of Pulmonary and Critical Care, University of Florida. “This is the time to be proactive, to develop ways to stay safe on the front lines. I encourage everyone to be a part of HERO – and part of the solution.”
To participate in the HERO program, click here to join the HERO trial registry.